Introduction to Breast Cancer
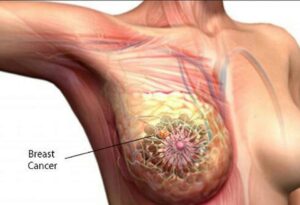
Breast cancer is a malignant tumor that originates in the cells of the breast. It is one of the most common cancers among women worldwide, though it can also occur in men. Understanding this complex disease is crucial for early detection, effective treatment, and improved patient outcomes. The breast is composed of lobules (glands that produce milk), ducts (tubes that carry milk to the nipple), and fatty and fibrous connective tissue. Breast cancer typically begins in the cells lining the milk ducts (ductal carcinoma) or the lobules (lobular carcinoma).
Global Prevalence and Statistics
Breast cancer remains a significant global health challenge. According to the World Health Organization (WHO), it is the most common cancer among women globally, accounting for 1 in 8 cancer diagnoses.
- Incidence Rates: In 2020, there were 2.3 million women diagnosed with breast cancer and 685,000 deaths globally. This makes it the most prevalent cancer worldwide in terms of new cases.
- Geographical Variations: Incidence rates vary significantly across different regions. Developed countries generally have higher incidence rates, often attributed to lifestyle factors, later childbearing, and increased screening. However, mortality rates are often higher in lower-income countries due to limited access to early diagnosis and effective treatment.
- Trends: While incidence rates have generally been rising, particularly in developing nations, advancements in screening and treatment have led to a decline in mortality rates in many high-income countries over the past few decades. This highlights the critical role of early detection and access to quality care.
Causes and Risk Factors
The exact cause of breast cancer is not fully understood, but a combination of genetic, environmental, and lifestyle factors is believed to contribute to its development.
Non-Modifiable Risk Factors:
- Gender: Being female is the primary risk factor. While men can get breast cancer, it is about 100 times more common in women.
- Age: The risk of breast cancer increases with age, with most cases diagnosed in women over 50.
- Genetics: Approximately 5-10% of breast cancers are hereditary, meaning they are caused by inherited gene mutations. The most common mutated genes are BRCA1 and BRCA2, which significantly increase the risk of breast and ovarian cancer. Other genes like PALB2, CHEK2, and ATM also play a role.
- Personal History of Breast Cancer: Women who have had breast cancer in one breast are at higher risk of developing it in the other breast or a recurrence in the same breast.
- Family History: Having a first-degree relative (mother, sister, daughter) with breast cancer, especially at a young age, increases the risk.
- Race/Ethnicity: White women have a slightly higher incidence of breast cancer overall, but Black women are more likely to be diagnosed with aggressive subtypes like triple-negative breast cancer and often have poorer outcomes.
- Dense Breast Tissue: Women with dense breasts (more connective tissue than fatty tissue) have a higher risk of breast cancer, and dense tissue can also make mammograms harder to interpret.
- Certain Benign Breast Conditions: Some non-cancerous breast conditions, such as atypical hyperplasia, can increase breast cancer risk.
Modifiable Risk Factors (Lifestyle and Environmental):
- Alcohol Consumption: Regular consumption of alcohol increases the risk of breast cancer, with the risk increasing with the amount of alcohol consumed.
- Obesity: Being overweight or obese, especially after menopause, increases breast cancer risk. Fat cells produce estrogen, which can fuel the growth of hormone-sensitive breast cancers.
- Physical Inactivity: A sedentary lifestyle is associated with an increased risk of breast cancer. Regular physical activity can help lower the risk.
- Hormone Therapy After Menopause: Combined hormone therapy (estrogen and progestin) used to manage menopausal symptoms can increase breast cancer risk. Estrogen-only therapy may also increase risk, though to a lesser extent.
- Reproductive History:
- Childbearing: Women who have never had a full-term pregnancy or had their first full-term pregnancy after age 30 have a slightly higher risk.
- Breastfeeding: Breastfeeding for a prolonged period may slightly lower breast cancer risk.
- Oral Contraceptives: Some studies suggest a very slight increase in breast cancer risk with the use of oral contraceptives, but this risk appears to revert to normal after stopping use.
- Radiation Exposure: Exposure to radiation, particularly to the chest area at a young age (e.g., for Hodgkin lymphoma treatment), can increase breast cancer risk.
- Diet: While no specific diet has been definitively proven to cause or prevent breast cancer, a diet high in processed foods, red meat, and saturated fats and low in fruits, vegetables, and whole grains may contribute to an increased risk.

Diagnosis and Staging
Early diagnosis is crucial for successful breast cancer treatment.
Diagnostic Methods:
- Clinical Breast Exam (CBE): A physical examination of the breasts by a healthcare professional to check for lumps or other changes.
- Mammogram: An X-ray of the breast, the primary screening tool for breast cancer. It can detect tumors before they are felt.
- Breast Ultrasound: Uses sound waves to produce images of breast tissue. Often used to further evaluate abnormalities found on mammograms or clinical exams, especially for dense breasts.
- Breast MRI (Magnetic Resonance Imaging): Uses magnets and radio waves to create detailed images of the breast. Often used for high-risk women, for staging purposes, or to assess the extent of cancer.
- Biopsy: The definitive diagnostic test. A small sample of breast tissue is removed and examined under a microscope by a pathologist to confirm the presence of cancer cells and determine their characteristics (e.g., hormone receptor status). Types include fine needle aspiration (FNA), core needle biopsy, and surgical biopsy.
Staging:
Once cancer is diagnosed, staging determines the size of the tumor, whether it has spread to lymph nodes, and if it has metastasized to distant parts of the body. This helps guide treatment decisions. The most common staging system is the TNM system (Tumor, Node, Metastasis).
- T (Tumor): Refers to the size of the primary tumor.
- N (Nodes): Indicates whether cancer has spread to nearby lymph nodes.
- M (Metastasis): Determines if the cancer has spread to distant organs (e.g., bones, lungs, liver, brain).
Based on these factors, breast cancer is typically staged from 0 to IV:
- Stage 0 (DCIS – Ductal Carcinoma In Situ): Non-invasive cancer confined to the milk ducts.
- Stage I: Small invasive cancer, usually less than 2 cm, with no lymph node involvement or very minimal involvement.
- Stage II: Larger tumor (2-5 cm) or spread to 1-3 axillary lymph nodes.
- Stage III (Locally Advanced): Larger tumor (over 5 cm), extensive lymph node involvement (4-9 lymph nodes), or spread to chest wall/skin.
- Stage IV (Metastatic): Cancer has spread to distant organs.
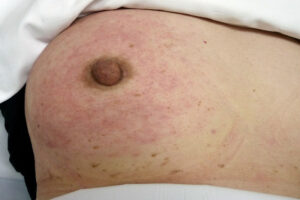
Treatment Approaches
Breast cancer treatment is highly individualized, depending on the stage, hormone receptor status (estrogen receptor (ER), progesterone receptor (PR)), HER2 status, and other tumor characteristics, as well as the patient’s overall health and preferences. Treatment often involves a combination of modalities.
Local Treatments:
- Surgery: The primary treatment for most breast cancers.
- Lumpectomy (Breast-Conserving Surgery): Removal of the tumor and a small margin of healthy tissue, preserving most of the breast. Usually followed by radiation therapy.
- Mastectomy: Removal of the entire breast. In some cases, a double mastectomy (removal of both breasts) may be performed, especially for women with a high genetic risk.
- Lymph Node Dissection:
- Sentinel Lymph Node Biopsy (SLNB): A procedure to remove only the first few lymph nodes that drain from the tumor. If these are cancer-free, no further lymph nodes are removed.
- Axillary Lymph Node Dissection (ALND): Removal of a larger number of lymph nodes in the armpit if cancer is found in the sentinel nodes.
- Radiation Therapy: Uses high-energy rays to kill cancer cells. Often given after lumpectomy to reduce the risk of recurrence, or after mastectomy in some cases, especially if lymph nodes are involved.
- External Beam Radiation: The most common type, delivered by a machine outside the body.
- Brachytherapy (Internal Radiation): Radioactive seeds are placed inside the breast for a short period.
Systemic Treatments (Targeting Cancer Cells Throughout the Body):
- Chemotherapy: Uses drugs to kill rapidly dividing cancer cells. Can be given before surgery (neoadjuvant chemotherapy) to shrink large tumors, after surgery (adjuvant chemotherapy) to kill remaining cancer cells, or for metastatic disease.
- Hormone Therapy (Endocrine Therapy): Used for hormone receptor-positive breast cancers (ER+ and/or PR+). These therapies block the effects of estrogen or lower estrogen levels, thereby starving the cancer cells.
- Tamoxifen: Blocks estrogen receptors in breast cancer cells. Used in pre- and post-menopausal women.
- Aromatase Inhibitors (AIs) (e.g., Anastrozole, Letrozole, Exemestane): Block the enzyme aromatase, which converts androgens into estrogen. Used primarily in post-menopausal women.
- Ovarian Suppression/Ablation: For pre-menopausal women, methods to stop the ovaries from producing estrogen (e.g., LHRH agonists, surgical removal of ovaries).
- Fulvestrant: An estrogen receptor downregulator, used for advanced hormone receptor-positive breast cancer.
- Targeted Therapy: Drugs that target specific molecules involved in cancer growth and progression, often with fewer side effects than chemotherapy.
- HER2-Targeted Therapies (e.g., Trastuzumab (Herceptin), Pertuzumab (Perjeta), Lapatinib, Kadcyla): Used for HER2-positive breast cancers, which have too many copies of the HER2 protein.
- CDK4/6 Inhibitors (e.g., Palbociclib, Ribociclib, Abemaciclib): Used in combination with hormone therapy for hormone receptor-positive, HER2-negative advanced or metastatic breast cancer. These drugs block enzymes that promote cell division.
- PARP Inhibitors (e.g., Olaparib, Talazoparib): Used for HER2-negative breast cancers with BRCA gene mutations. These drugs interfere with DNA repair in cancer cells.
- PI3K Inhibitors (e.g., Alpelisib): Used for hormone receptor-positive, HER2-negative advanced breast cancer with a PIK3CA mutation.
- Immunotherapy: Helps the body’s immune system fight cancer.
- PD-1/PD-L1 Inhibitors (e.g., Pembrolizumab, Atezolizumab): Used for certain types of advanced triple-negative breast cancer (TNBC) that express PD-L1. These drugs block proteins that prevent immune cells from attacking cancer cells.
Latest Research and Future Outlook
The field of breast cancer research is rapidly evolving, bringing new hope for improved outcomes.
- Precision Medicine and Genomic Profiling:
- Enhanced Genomic Testing: Beyond BRCA, comprehensive genomic profiling of tumors is becoming standard. This helps identify specific mutations and biomarkers that can be targeted by specific drugs, leading to truly personalized treatment plans.
- Liquid Biopsies: Analyzing circulating tumor DNA (ctDNA) in blood samples can provide real-time information about tumor mutations, treatment response, and minimal residual disease (MRD), potentially guiding treatment adjustments earlier and less invasively.
- Artificial Intelligence (AI) and Machine Learning: AI is being used to analyze vast amounts of patient data, identify patterns, predict treatment response, and even assist in image interpretation for early detection, potentially leading to more accurate diagnoses and personalized risk assessments.
- Advanced Targeted Therapies:
- Antibody-Drug Conjugates (ADCs): These are “smart bombs” that combine a targeted antibody with a chemotherapy drug. The antibody delivers the drug directly to cancer cells, minimizing damage to healthy cells. Trastuzumab deruxtecan (Enhertu) is a prime example showing remarkable efficacy in HER2-low and HER2-positive breast cancer. More ADCs targeting other proteins are in various stages of development.
- Novel Kinase Inhibitors: New inhibitors targeting various signaling pathways involved in cancer cell growth and survival are continuously being developed, broadening the spectrum of targeted treatments.
- Expanding Immunotherapy Applications:
- Combination Immunotherapy: Researchers are exploring combinations of different immunotherapy drugs or immunotherapy with chemotherapy, targeted therapy, or radiation to enhance the immune response against breast cancer, particularly for less responsive subtypes like ER+ breast cancer.
- Cellular Immunotherapy: CAR T-cell therapy, while currently more established in blood cancers, is being investigated for solid tumors like breast cancer. This involves genetically modifying a patient’s own T-cells to recognize and attack cancer cells.
- Vaccines: Therapeutic vaccines aimed at stimulating the immune system to recognize and destroy breast cancer cells are under investigation, both for preventing recurrence and treating advanced disease.
- Drug Repurposing and Synergistic Combinations:
- Repurposing Existing Drugs: Investigating whether existing drugs approved for other conditions could be effective against breast cancer, potentially accelerating drug development and reducing costs.
- Synergistic Drug Combinations: Identifying drug combinations that work better together than individually, often by targeting multiple pathways or overcoming resistance mechanisms.
- Prevention Strategies:
- Chemoprevention: Further research into medications like tamoxifen and aromatase inhibitors for reducing breast cancer risk in high-risk individuals.
- Lifestyle Interventions: Continued emphasis on large-scale studies to confirm the impact of diet, exercise, and weight management on breast cancer risk and recurrence, leading to more robust evidence-based prevention guidelines.
- Addressing Disparities:
- Equity in Access: Efforts to reduce disparities in breast cancer outcomes by improving access to screening, diagnosis, and treatment in underserved populations globally.
- Tailored Approaches: Research into understanding biological and social factors contributing to worse outcomes in certain racial/ethnic groups and developing tailored interventions.
Conclusion
Breast cancer research has made remarkable strides, transforming it from a universally fatal disease to one with increasingly high survival rates, especially when detected early. The future of breast cancer treatment lies in even greater personalization, driven by advanced genomic insights, innovative targeted therapies, and the growing power of immunotherapy. While challenges remain, the continuous pursuit of scientific discovery offers immense hope for even more effective, less toxic treatments and, ultimately, a future where breast cancer is preventable and curable for all. Regular screenings, awareness of risk factors, and supporting research are key to continued progress in the fight against this disease.